|
Here, depending of the density of feed or solvent we decide which stream will enter from the top. 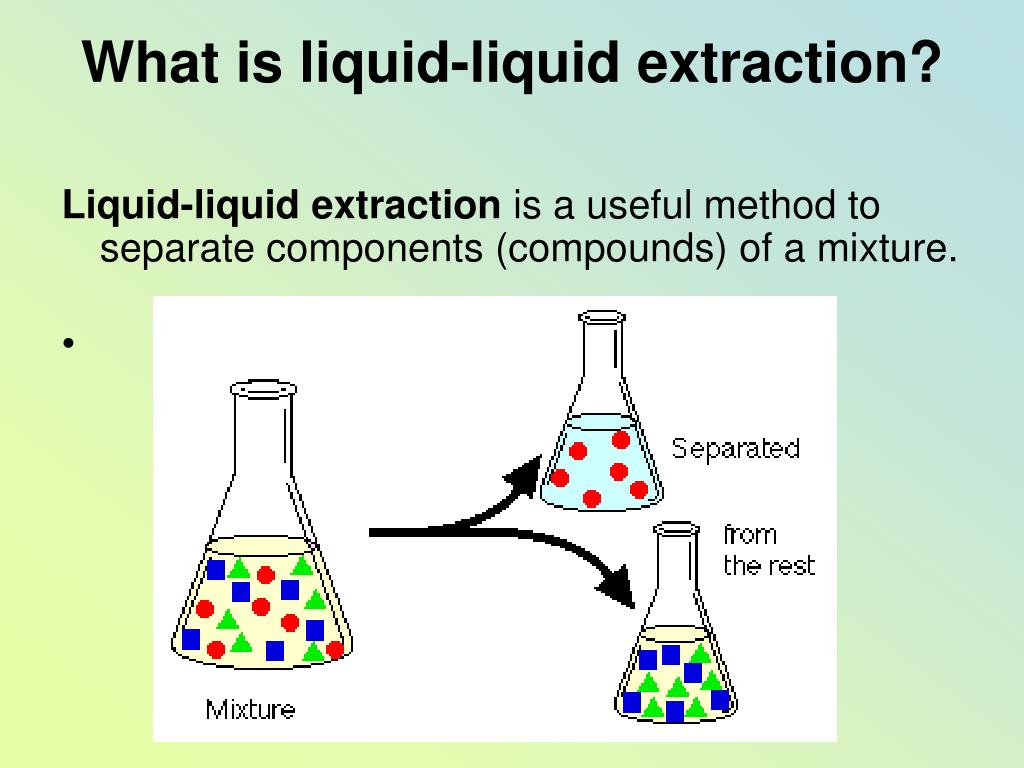
Below figure is for the reference to understand an extraction column process. Process of Continuous Extraction Columnįor efficient operation of the extraction column you should provide precise interface control. Conversely, when light phase is in continuous phase, we maintain interface at the bottom side of the extractor. If light phase is in disperse form then interface is maintained at top side of the extraction column. The phase which is in form of droplets inside extractor is known as disperse phase and other is called continuous phase. The function of column internals is to form fine droplet of one phase into other phase to enhance solute mass transfer. Mechanically agitated type extractors are known as Rotating Disc Contact type extractor also. Inside extraction columns mechanical agitation, trays or packing is used to provide the surface are for solute mass transfer. Light phase is fed from the bottom side and heavy phase is fed from top side of the extraction column. The hydraulics of extractor column is established by density difference of both phases. Continuous Liquid – Liquid Extraction System This problem depends on process temperature and presence of inert solids. Emulsion is the state in which there is no differentiation between two phases. In case of emulsion formation phase separation becomes difficult. Where, μ is the viscosity (cP) of continuous phase, ρ f & ρ s are the densities (kg/m3) for feed & solvent respectively. The settling time for the phases you can estimate by below equation The solute rich layer is our extract phase and the other layer is raffinate phase. Further these layers are collected in different vessels. After required settling time, which depends on the density difference and viscosity of continuous phase, two distinct layers form. 
After mixing when equilibrium is achieved between phases mixing is stopped and mass is allowed to settle down. In batch extraction systems we use agitated vessel to mix feed and solvent streams. So, conceptually we can visualize an extraction process is the combination of two process operations. Process Calculations for Single Stage Continuous Extraction System.Process of Continuous Extraction Column.Continuous Liquid – Liquid Extraction System.In case of change in densities K may not be constant. 
Since, C A I = ρ I*y A and C A II = ρ II*x A, Therefore, we can write phase equilibrium constant in terms of mass fraction as below Where, M is distribution coefficient, C A Iand C A II are the solute concentration (g/liter) at equilibrium state in Phase- I and Phase- II respectively. The ratio of solute concentration at equilibrium state between liquid Phase- I and liquid Phase- II is known as distribution coefficient and given by below expression. In which upper layer is light phase and bottom layer being heavy phase. After mixing when this mixture settles, two layers forms. In extraction process feed and solvent streams mixing enhance the solute transfer from feed to solvent stream. Selection of solvent for extraction is based on many criteria such as boiling point, environment friendly, cost and toxicity. And, other is solvent stream in which solute transfer takes place. However, the solvent which contain solute we consider it a feed stream. Moreover, both the solvents are insoluble in one another. In liquid-liquid extraction process solute is transferred between solvents. Where the subscripts indicate the extraction number with 0 representing the system before the extraction and 1 the system following the first extraction.In plants liquid-liquid extraction column is used to separate components using solvent.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
